Research
Project for immune responses in infectious diseases
Infectious diseases by viruses, such as COVID-19, Influenza, viral hepatitis and AIDS, are great threats of our daily lives. In order to defeat them, it must be important to understand our immune system. We have two highly organized immune machineries, innate and adaptive immunity. Among them, innate immunity is critical for not only swift eradication of the infected pathogens but also fine tuning of the subsequent adaptive immunity. In 2004, we identified cytoplasmic viral RNA sensor molecules, RIG-I-like receptors (RLRs), and demonstrated their essential function in anti-viral innate immunity. In this project, we are analyzing physiological significance and molecular mechanism of RLRs-mediated anti-viral signaling.
LEARN MORE
The summary of our research results is as follows:
- RLRs play a critical role in innate antiviral immunity, including activation of type I interferon genes‘.
- Among three RLR family members (RIG-I, MDA5, and LGP2), RIG-I and MDA5 detect viral non-self RNA structures with distinct specificities (i.e., 5‘triphosphate-containing short double-stranded RNA (dsRNA) and long dsRNA, respectively).
- In response to viral non-self RNA, RIG-I and MDA5 switch their conformation in an ATP-dependent manner and transmit the signal via caspase recruitment domain (CARD)-CARD interaction with the downstream mitochondrial adaptor molecule, IPS-1/MAVS.
- RLRs recognize substrate RNA with C-terminal negatively charged domain (CTD).
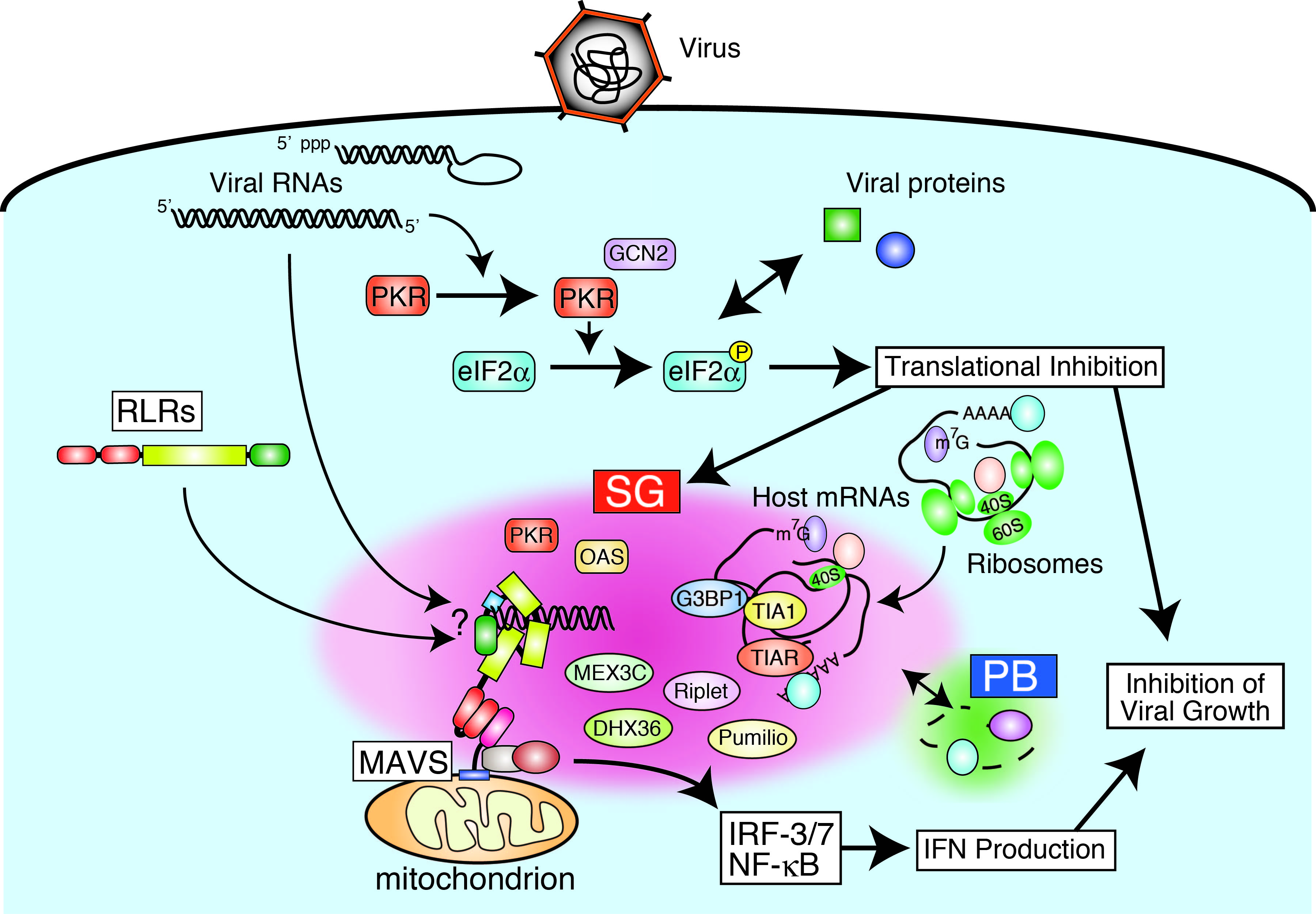
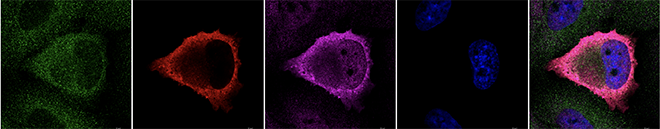
- RLRs localize stress granule (SG)-like structure in response to viral infection, suggesting a possible requirement of SG for RLR-mediated signal activation.
In this project, we aim to elucidate the physiological significance of RLRs in innate antiviral immunity and the molecular mechanisms underlying the detection of viral non-self RNA and signal transduction by RLRs. Since it is still unclear whether RLRs are recruited to SGs and involved in detecting the foreign RNAs in SGs, we are analyzing their molecular mechanism and function. We are also investigating whether RLRs can interact with endogenous self-RNAs and have unknown biological functions. Furthermore, since the function of RLRs is associated with diseases including autoinflammation and COVID-19, we are analyzing the function of RLRs using molecular, biochemical, cell biological approaches, and mouse models.